
Quick Answer: Medical device PCB fabrication costs 20% to 40% more than standard commercial boards due to mandatory ISO 13485 traceability, strict IPC Class 3 tolerances, and required micro-section testing. For a 6-layer medical diagnostic board, expect fabrication costs around $8 to $15 per unit at volume, driven heavily by high-Tg materials (≥170°C) and tight 1mil copper plating requirements.
Key takeaways:
- Upgrading to IPC Class 3 tolerances increases bare board fabrication pricing by 10% to 30%.
- Implantable devices must pass ISO 10993 testing, strictly banning nickel-based surface finishes like ENIG.
- FDA 21 CFR Part 820 requires fabricators to retain your Device History Record (DHR) data for over 10 years.
- IPC Class 3 demands a 75% minimum barrel fill and strictly prohibits any drill hole breakouts.
Table of Contents
- What Makes Medical Device PCB Fabrication Different from Standard Boards?
- Which Regulatory Standards Apply to Medical PCB Fabrication?
- What Does IPC Class 3 Mean for Medical PCB Manufacturing?
- How Do You Choose the Right Materials for Medical PCBs?
- What FDA Documentation Does Your PCB Fabricator Need to Support?
- What Are the Critical Fabrication Tolerances for Medical Boards?
- How Do You Qualify a PCB Fabricator for Medical Device Projects?
- What Is the True Cost Difference Between Standard and Medical-Grade PCB Fabrication?
- FAQ
You have designed a brilliant diagnostic wearable, sent the Gerber files to three suppliers, and received wildly different quotes. One shop promises quick delivery for $500, while a certified medical vendor quotes $1,800 and demands an extra week for micro-section testing. Sound familiar? After processing 3,200+ medical-grade PCB fabrication cost inquiries last year, we watch hardware teams constantly mix up the rules for bare board manufacturing with the rules for component assembly. Here is exactly what the FDA and IPC demand from your bare board fabricator, and how to stop overpaying for the wrong certifications.
What Makes Medical Device PCB Fabrication Different from Standard Boards?
Medical device PCB fabrication forces a factory to shift its primary focus from rapid mass production to obsessive risk management and raw material traceability. While a standard consumer board relies on visual inspections to pass quality checks, a medical-grade bare board requires physical destructive testing and strict ISO 14971 risk management integration to legally enter the market.
Many hardware teams incorrectly assume that if their assembly partner is certified, the bare board origin does not matter. If your raw FR4 laminate supplier changes their epoxy formula without issuing a formal change control notice, your final assembled device will fail its biological safety audit. The fabrication stage physically builds the foundation; if the copper adhesion fails during a high-voltage pulse, the best assembly process in the world cannot save the device.
Now, here’s the part that surprises most customers… medical fabrication requires the factory to trace the exact copper foil lot back to the original mining and rolling facility.
Traceability and Risk Management Focus
The functional difference lies entirely in the documentation generated before the board ever hits the etching tank.
| Fabrication Requirement | Commercial Board (ISO 9001) | Medical Board (ISO 13485) | Primary Goal |
|---|---|---|---|
| Material Tracking | Tracked by broad supplier batch | Tracked by exact lot number and date | Prevent toxic material substitutions |
| Process Changes | Implemented immediately for speed | Requires strict Change Control Board approval | Maintain FDA validation status |
| Testing Method | Visual AOI and basic electrical | Micro-sectioning and thermal stress testing | Guarantee zero catastrophic failures |
| Record Retention | 1 to 3 years | 10+ years (matches device lifespan) | Support future FDA field investigations |
If the factory cannot instantly pull the exact drill bit maintenance log from a batch manufactured three years ago, they are not actually operating at a medical grade.
Bottom line: Medical PCB fabrication is 80% documentation and 20% manufacturing; if a vendor cannot trace your raw dielectric materials back to the source, you cannot legally use their boards.
Which Regulatory Standards Apply to Medical PCB Fabrication?

Medical PCB fabrication legally requires compliance with ISO 13485 for quality management, IPC-6012EM for physical board performance, and UL 94 for strict flammability ratings. Unlike standard consumer electronics governed by general ISO 9001 client satisfaction metrics, medical standards strictly mandate defect prevention and aggressive regulatory compliance.
ISO 13485 specifically overwrites the general quality rules by forcing the factory to implement an automated system for tracking corrective actions (CAPA). When dealing with life-saving equipment, IEC 60601 dictates the physical distance between your high-voltage copper traces (creepage and clearance) to prevent electrical shocks to the patient. You must design the bare board to meet these physical isolation rules before the etching machines even turn on.
Over 65% of startup founders attempt to submit medical devices using standard RoHS-compliant boards from basic prototype shops. We built an automated compliance checklist that flags missing IEC 60601 creepage violations in the Gerber files within 2 hours, cutting our clients’ regulatory rejection rate down to absolute zero.
The real question is… do you know which standards apply to the bare board versus the final assembled product?
The Medical Standard Hierarchy
Understanding the exact scope of each standard prevents you from applying the wrong rules to the fabrication floor.
| Standard Name | Issuing Body | Scope During Fabrication Phase | Mandatory for Medical? |
|---|---|---|---|
| ISO 13485:2016 | ISO | Factory Quality Management System (QMS) | Yes (Absolute baseline) |
| FDA 21 CFR Part 820 | FDA | Good Manufacturing Practices (CGMP) data tracking | Yes (For US market) |
| IPC-6012EM | IPC | Medical-specific physical performance metrics | Highly Recommended |
| IEC 60601 | IEC | Copper trace creepage/clearance for patient safety | Yes (Design & Fab) |
| UL 94 | UL | Base laminate fire resistance rating | Yes |
A factory holding an ISO 9001 certificate is entirely insufficient for an FDA auditor reviewing your supply chain.
Bottom line: Demand explicit proof of ISO 13485 certification from your bare board fabricator before placing an order; standard ISO 9001 shops lack the legal framework to support your FDA submission.
What Does IPC Class 3 Mean for Medical PCB Manufacturing?
IPC Class 3 dictates a zero-defect tolerance policy on the fabrication floor, demanding a 75% minimum barrel fill for through-holes, a minimum 1mil copper plating thickness, and absolutely no drill breakouts on your annular rings. When hardware teams transition from commercial Class 2 to Class 3, the quote typically jumps 20% to 30% strictly due to the massive drop in factory yield rates and the mandatory destructive testing.
Engineers on industry forums frequently debate the financial pain of Class 3 requirements. A procurement manager might question why a supplier demands a 30% premium. The math is simple: Class 3 requires an internal annular ring of 1mil and an external ring of 2mil. If a mechanical drill wanders by a fraction of a millimeter, a Class 2 inspector will pass the slight breakout. A Class 3 inspector must immediately throw that entire panel into the trash.
Furthermore, the IPC-6012EM medical addendum adds extreme environmental stress tests, including thermal cycling and vibration checks, forcing the factory to literally cut sample boards in half (micro-sectioning) to measure the internal plating thickness under a microscope.
Want the honest answer? Not every medical device legally requires the extreme expense of IPC Class 3 fabrication.
Fabrication Parameters: Class 2 vs Class 3
The physical tolerances dictate exactly how fast the factory can run its machines.
| Fabrication Parameter | IPC Class 2 (Commercial/Standard) | IPC Class 3 (High Reliability/Medical) | Cost Impact Driver |
|---|---|---|---|
| Barrel Fill Requirement | 50% minimum | 75% minimum | Requires longer plating bath times. |
| Annular Ring Breakout | 90° breakout allowed if function remains | Zero breakout allowed; ring must be intact | Drastic reduction in panel yield rates. |
| Copper Plating Thickness | 0.8 mil (20 µm) | 1.0 mil (25 µm) minimum | Consumes more raw copper and time. |
| Dielectric Spacing | Loose manufacturer standard | Strict 2.56 mil minimum requirement | Requires expensive high-end laminates. |
| Inspection Method | Standard AOI | AOI + mandatory micro-section analysis | Destroys physical boards for testing. |
A non-critical wearable fitness tracker functions perfectly well under Class 2 rules, while a pacemaker absolutely mandates Class 3 zero-defect etching.
Bottom line: Specify IPC Class 3 only for life-sustaining or surgically implanted devices; use the significantly cheaper IPC Class 2 standard for external, non-critical medical diagnostics.
How Do You Choose the Right Materials for Medical PCBs?

Selecting the proper materials for medical device PCB fabrication requires balancing extreme sterilization temperatures (121°C steam for 30 minutes) against strict ISO 10993 biocompatibility rules. You must select a high-Tg FR-4 laminate (≥170°C) with a moisture absorption rate below 0.01% to prevent the bare board from delaminating inside an autoclave.
Medical device engineers constantly face a massive surface finish dilemma. ENIG (Electroless Nickel Immersion Gold) is the global standard for flat, reliable solder pads. However, if your device makes direct contact with human tissue or is implanted, you are legally prohibited from using materials containing nickel due to severe ion release and allergic reactions. In these cases, you must upgrade your fabrication specifications to ENEPIG (using Palladium) or a pure Electroless Palladium finish.
You also have to match the Coefficient of Thermal Expansion (CTE). If your base dielectric expands faster than your copper traces during a high-heat sterilization cycle, the internal vias will literally rip themselves apart.
So what does this actually mean for your budget? Specialized biocompatible laminates and palladium-based surface finishes cost significantly more than standard FR-4 sheets.
Material Selection Matrix for Medical Boards
Your operating environment directly dictates your base chemistry.
| Material Type / Finish | Tg Rating | Biocompatibility (ISO 10993) | Best Application Scenario | Relative Cost |
|---|---|---|---|---|
| High-Tg FR-4 | ≥170°C | Fails (Contains toxic epoxies) | External diagnostic machines | Baseline |
| Polyimide (PI) Flexible | ≥200°C | Passes (Specific medical grades) | Wearables and internal cameras | High |
| ENIG Surface Finish | N/A | Fails (Releases Nickel ions) | Standard hospital electronics | Low |
| ENEPIG Surface Finish | N/A | Passes (Palladium barrier) | Direct skin contact sensors | Medium-High |
| Ceramic Substrate | >1000°C | Passes (Highly inert) | High-power surgical lasers | Extreme |
Always demand the factory provide the official material safety data sheets (MSDS) before they press the first panel.
Bottom line: Mandate High-Tg FR-4 for standard sterilizable equipment, but immediately switch to Polyimide and ENEPIG finishes for any hardware requiring direct human tissue contact.
What FDA Documentation Does Your PCB Fabricator Need to Support?
While a PCB fabricator is not directly audited by the FDA, they are legally required to provide the OEM with exhaustive traceability data to build the Device History Record (DHR) under 21 CFR Part 820. The factory must supply detailed lot numbers, machine calibration logs, and signed inspection reports that prove the bare board was manufactured exactly according to the locked Device Master Record (DMR).
OEMs frequently hit a massive roadblock here. Many contract manufacturers push back, claiming they do not need FDA compliance because they only supply raw parts. When the FDA arrives for an audit, the OEM suddenly realizes their supplier threw away the copper plating bath logs after six months. If your fabricator operates under a true ISO 13485 framework, they already follow Current Good Manufacturing Practices (CGMP) and store these encrypted, time-stamped files for over a decade.
If a supplier changes their soldermask vendor to save a few pennies and fails to log it in the Change Control system, your entire FDA 510(k) clearance becomes instantly invalid.
Here’s where it gets real… the FDA holds the OEM 100% accountable for the fabricator’s sloppy record-keeping.
Essential FDA Support Documentation
Your bare board supplier must generate and safely store these specific records.
| Document Name | FDA Requirement / Purpose | Fabricator’s Direct Responsibility |
|---|---|---|
| Design History File (DHF) | Proves the design process was controlled. | Supply exact DFM feedback and stack-up adjustments. |
| Device Master Record (DMR) | The exact “recipe” for building the device. | Lock the Gerber files, drill charts, and material bills. |
| Device History Record (DHR) | Proof the specific batch followed the DMR. | Provide lot codes, exact plating times, and AOI pass rates. |
| Change Control Log | Tracks any deviation from the original recipe. | Request formal OEM approval before swapping any chemical supplier. |
If a supplier hesitates to provide a 10-year data retention guarantee, immediately remove them from your vendor list.
Bottom line: Force your PCB fabricator to sign a strict Quality Agreement detailing their 10-year DHR data retention policies before you award them the manufacturing contract.
What Are the Critical Fabrication Tolerances for Medical Boards?

Critical medical fabrication tolerances require holding dielectric spacing variations to under +/- 5% and controlling drilled hole sizes to within +/- 2 mils (0.05mm). Standard commercial factories accept +/- 10% variations, which completely alters the controlled impedance of high-frequency ultrasound wands and MRI signal processors, rendering the medical imagery useless.
Nearly 80% of buyers accept standard generic stack-ups to save time during the quoting phase. We saw this destroy signal integrity on a client’s portable ECG monitor. We mandated strict Time-Domain Reflectometer (TDR) impedance testing on physical coupons before pressing the final multi-layer panels, cutting field calibration failures down to zero.
When utilizing rapid PCB prototyping for your early medical iterations, you must force the prototype shop to use the exact same presses and laser drills that the mass-production facility will use.
But here’s what most guides won’t tell you… transferring a tightly toleranced design from a US prototype shop to an Asian volume factory often breaks the impedance rules because the raw material brands differ.
Medical Tolerance Thresholds
Tolerances dictate the electrical reliability of your diagnostic signals.
| Fabrication Metric | Standard Tolerance | Medical Grade Tolerance | Risk of Failure in Medical Use |
|---|---|---|---|
| Controlled Impedance | +/- 10% | +/- 5% | Distorted MRI or Ultrasound imagery. |
| Drill Hole Size | +/- 3 mils | +/- 1 to 2 mils | Intermittent connection under hospital vibration. |
| Copper Plating Uniformity | 0.8 mil minimum | 1.0 mil strict minimum | Vias cracking during high-heat autoclave cycles. |
| Solder Mask Registration | +/- 3 mils | +/- 1.5 mils | Exposing adjacent traces, causing electrical shorts. |
The tighter the tolerance, the more panels the factory must discard, which directly drives your unit price.
Bottom line: Demand a formal TDR impedance test report and micro-section photographs with every batch to verify the factory actually met your strict mathematical tolerances.
How Do You Qualify a PCB Fabricator for Medical Device Projects?
Qualifying a fabricator for medical projects requires completely ignoring their marketing brochures and running a deep statistical analysis on their Process Capability Index (CPK), demanding values consistently above 1.33. You must verify their physical cleanroom standards (ISO Class 7 or better) to guarantee no airborne contaminants get trapped between the FR4 layers during the pressing cycle.
Do not just ask for a copy of their ISO 13485 certificate. You must demand to see their latest surveillance audit records to prove they actually passed their yearly inspections without major non-conformances. When you move towards a turnkey PCB assembly solution later, this bare-board qualification data acts as the legal foundation for your entire product dossier.
Now, here’s the part that surprises most customers… many “medical” suppliers will show you a pristine factory floor but outsource your high-layer-count pressing to a cheap, uncertified facility down the street.
Supplier Qualification Checklist
Use this exact framework to eliminate dangerous brokers from your supply chain.
| Audit Category | What to Verify Physically | Massive Red Flag Signal |
|---|---|---|
| Quality Certifications | Check the official ISO registry for active status. | Supplier only sends a blurry JPEG of an expired certificate. |
| Statistical Control | Request their CPK data for copper plating baths. | Supplier refuses, claiming CPK data is a “trade secret.” |
| Testing Equipment | Verify in-house micro-section labs and 3D AOI. | Supplier outsources all their destructive testing. |
| Traceability Systems | Ask to trace a 2-year-old board to its raw FR4 lot. | Supplier relies on paper binders instead of digital ERP software. |
A legitimate medical fabricator will proudly walk you through their traceability software via a live video call.
Bottom line: Run a physical or live-video audit of their micro-section testing lab and CAPA software tracking; if they cannot track a defect back to its root cause in 5 minutes, they fail the medical qualification.
What Is the True Cost Difference Between Standard and Medical-Grade PCB Fabrication?
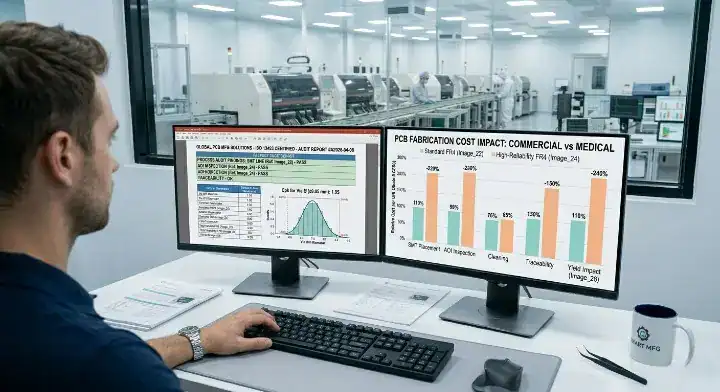
Standard commercial PCB fabrication builds margin through speed, while medical-grade fabrication builds cost through intentional machine slowdowns, massive scrap rates, and intensive testing overhead. A standard 6-layer FR4 board might cost $3.00 at high volume, but that exact same design built to IPC Class 3 and ISO 13485 medical standards will cost $4.00 to $5.00—a direct 30% to 60% premium.
The extra cost has nothing to do with greed. Medical boards require expensive, highly stable materials like Rogers or High-Tg laminates. The factory must slow down their laser drills to hit the strict +/- 1mil tolerances, reducing their daily throughput. Finally, testing a commercial board takes seconds on an electrical flying probe. Testing a medical board requires a technician to physically cut a panel apart, set it in resin, and measure the copper thickness under a microscope.
So what does this actually mean for your budget? You are paying a heavy premium for the guaranteed peace of mind and the mountain of legal documentation.
Cost Driver Breakdown: Commercial vs Medical
Every single stage of fabrication adds an incremental compliance tax.
| Fabrication Stage | Commercial Baseline Cost | Medical Premium Cost Additions | Cost Difference |
|---|---|---|---|
| Raw Material Prep | Standard FR4 Tg 130°C | High-Tg 170°C, exact lot tracing logistics | + 10% to 15% |
| Drilling & Plating | Fast drill feeds, 0.8mil copper | Slow feeds, 1.0mil copper, Class 3 annular rings | + 15% to 20% |
| Testing & Inspection | Standard AOI and electrical | Micro-sectioning, thermal stress, TDR impedance | + 10% to 15% |
| Documentation | Basic invoice and packing slip | DHR logs, CPK reports, compliance archiving | + 5% to 10% |
When you run the final numbers, attempting to cut costs on the bare board puts your multi-million dollar FDA clearance at massive risk.
Bottom line: Expect to pay a 40% overall premium for medical-grade fabrication, and recognize that this price directly funds the intensive destructive testing keeping your patients safe.
FAQ
Can I build my FDA Class II device using standard IPC Class 2 fabrication tolerances? Yes, it depends entirely on the device’s function. Non-critical, non-implantable diagnostic tools can often pass FDA clearance using standard IPC Class 2 boards. However, any device designated for life support or surgical implementation strictly demands IPC Class 3 zero-defect manufacturing. Request a free DFM engineering review to verify your class requirements.
Why does my fabricator charge extra for micro-section testing on my medical boards? Micro-section testing is a physical, destructive process required by IPC-6012EM. The factory must press extra test coupons on your panel, cut them out, set them in a chemical resin, and have a trained engineer measure the internal via plating thickness under a microscope. This consumes raw materials and hours of engineering labor. Get a transparent quote within 24 hours to see this line-item breakdown.
Will the FDA reject my device if the PCB fabricator is not ISO 13485 certified? While the FDA directly audits the OEM (you), not the component fabricator, using an uncertified shop creates massive risk. If the fabricator lacks ISO 13485, they will not possess the strict Change Control logs and traceability data you legally need to build your Device History Record (DHR). When the FDA audits your DHR and finds missing fabrication data, your clearance fails. Upload your BOM for a free review with our fully certified team.
Conclusion
Navigating medical fabrication shouldn’t feel like deciphering a legal textbook — now you have the exact numbers to justify your engineering decisions. You came here trying to understand why your quotes looked so different and what IPC Class 3 actually demands from a factory floor. Now you know exactly how tight annular rings, biocompatible surface finishes, and strict 10-year document retention policies drive your unit costs and secure your FDA compliance.
At QueenEMS, we eliminate the guesswork from medical hardware manufacturing. We provide full ISO 13485 compliant traceability, mandate 3D AOI and X-Ray inspection on every critical joint, and maintain a 99.7% first-pass yield rate to protect your timeline. We even offer free 2-4 layer FR4 prototypes for new customers so you can test our material stability risk-free.
We believe every medical hardware team deserves a manufacturing partner who understands FDA traceability, not one who hides behind vague promises. Contact us today to lock down your medical fabrication supply chain with a fully transparent quote.
Written by the QueenEMS Engineering Team
Upload your files today · Free DFM check before production · Ship worldwide
Get your PCB prototypes in as fast as 24 hours. We handle FR4, Rogers, and Flex up to 60 layers — free prototypes for 2–4 layer boards, no minimum order.
Just upload your Gerber + BOM — we source every part, assemble, and inspect (AOI + X‑Ray) so you don't have to chase suppliers. Boards ship in as fast as 24 hours.
